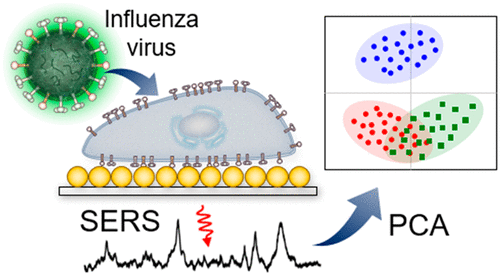
2004. Kwang-il Lim, Shakti Narayan, John A.T. Young, John Yin*. "Effects of lipid rafts on dyna

The association of cell surface receptors with sterol-sphingolipid-enriched microdomains of the plasma membrane, so-called lipid rafts, may affect the receptor-mediated entry and trafficking dynamics of viruses. A model retrovirus, subgroup A avian sarcoma and leukosis virus (ASLV-A), can initiate infection by binding to either of two forms of the tumor virus subgroup A (TVA) receptor, a lipid-raft-associated glycosylphosphatidylinositol (GPI)-anchored receptor (TVA800) or a transmembrane receptor (TVA950). Narayan et al. previously found that virus particles bound to TVA950 were more rapidly internalized than virions bound to TVA800, and the internalization via TVA950 exhibited biphasic kinetics. To explore potential molecular mechanisms for these results we developed a mathematical model that accounts for internalization of viruses through cellular pits, trafficking to an endosomal compartment where fusion occurs, and viral DNA synthesis. By fitting the model to experimental data we found that viruses bound to TVA950 were internalized up to 2.6-fold more rapidly than viruses bound to TVA800. Two- to threefold greater lateral diffusivities of transmembrane proteins, relative to GPI-anchored proteins, observed in other systems, suggest that the internalization rate of ASLV-A is diffusion-limited. Furthermore, by allowing for recycling of internalized TVA950-bound viruses back to the cell surface, we can account for the observed biphasic internalization kinetics. This mechanism is also consistent with the observed slower rate of DNA synthesis for viruses that enter via TVA950. Overall, the model provides a means to generate new experimentally testable hypotheses and sets a foundation for building a quantitative and integrated understanding of viral entry, trafficking, and intracellular dynamics.
StartFragment
PMID: 15137076 DOI: 10.1002/bit.20108
EndFragment